Learning how to inject peptides safely is a top priority for anyone looking to optimize their health, but navigating the “Wild West” of online suppliers can be overwhelming. While these powerful amino acid chains offer incredible benefits for recovery and focus, you shouldn’t have to play detective with your own wellness. True safety starts with a “Gold Standard” product and a sterile protocol. Let’s break down how to verify the purity of your supply and master the technique so you can feel confident in every step of your journey.
When it comes to your health, you shouldn’t have to play detective. Since these products often bypass the standard pharmacy shelf, the responsibility for quality control falls on us.
Here is your ultimate guide to ensuring the peptides you use are safe, pure, and effective.
How To Inject Petides Guide for Sterile and Painless Peptide Administration
When it comes to peptides, the injection technique is just as important as the quality of the product itself. Most therapeutic peptides are administered via subcutaneous injection—meaning they go into the fatty tissue just below the skin, not into the muscle.
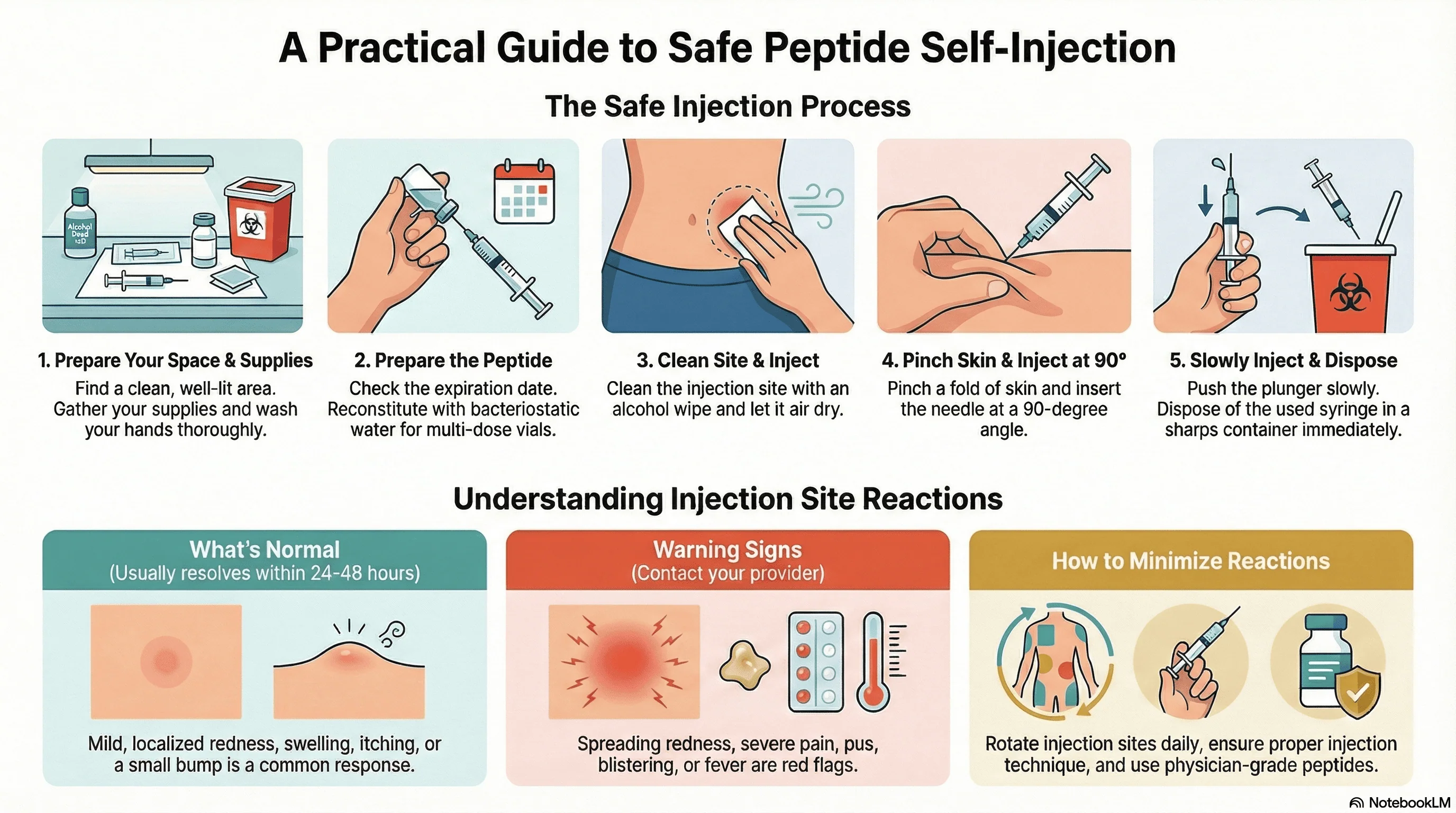
1. Prepare Your “Clean Zone” Before Injection
Before you even touch a vial, set up a dedicated workspace.
- Wash your hands thoroughly with soap and warm water for at least 20 seconds.
- Sanitize the surface where you’ll be working with an alcohol wipe.
- Gather your supplies: Your peptide vial, a fresh insulin syringe (usually 29–31 gauge), and two alcohol pads.
2. Prepare the Vial
- Swirl, Don’t Shake: If the peptide was recently mixed, gently swirl the vial. Never shake it, as peptides are fragile and can “break” (denature) under high friction.
- Sanitize the Stopper: Wipe the rubber top of the vial with a fresh alcohol pad and let it air dry. Do not blow on it to speed it up—this introduces bacteria from your breath.
3. Draw the Dose
- Air Pressure: Pull the syringe plunger back to the mark of your desired dose to fill it with air. Insert the needle into the vial and push the air in. This prevents a vacuum from forming, making the liquid easier to draw.
- The Draw: Invert the vial (upside down) and pull the plunger back slowly.
- Clear the Bubbles: Tap the side of the syringe to send any air bubbles to the top, then gently push the plunger to expel them. Small bubbles aren’t usually dangerous in fatty tissue, but they can make your dosing inaccurate.
4. The Injection Technique
The best sites are the abdomen (at least 2 inches away from the belly button) or the outer thigh.
- Sanitize the Skin: Use a fresh alcohol wipe on the injection site. Let it dry completely.
- The Pinch: Use your non-dominant hand to gently pinch about an inch of fatty tissue.
- The Angle: Insert the needle at a 45° to 90° angle. Because insulin needles are very short, a 90° (straight in) angle is standard for most people.
- Inject Slowly: Depress the plunger steadily. Once finished, wait about 5 seconds before pulling the needle out to ensure all the liquid stays in the tissue.
- No Rubbing: If there is a tiny drop of blood, apply light pressure with a cotton ball, but do not rub the area, as this can irritate the tissue.
5. Safe Disposal
- Never Reuse: Even one use dulls the needle (making the next shot hurt) and introduces a massive risk of infection.
- Sharps Container: Immediately place the used syringe in an FDA-cleared sharps container. If you don’t have one, a heavy-duty plastic laundry detergent bottle with a screw-on lid works in a pinch—just label it clearly.
Critical Safety Tip: Site Rotation
Never inject in the exact same spot twice in a row. This prevents lipohypertrophy (the buildup of hardened fatty tissue), which can stop your body from absorbing the peptide correctly.
1. The “Kitchen” Matters: Why GMP is Non-Negotiable
Think of Good Manufacturing Practices (GMP) as the Michelin Star of pharmaceutical production. It’s not just a suggestion; it’s a rigorous system that ensures every vial is produced in a clean, controlled environment.
- Regulatory Rigor: Look for manufacturers that follow FDA or EU standards. This means their facilities are regularly inspected, their equipment is calibrated, and their staff is highly trained.
- The Paper Trail: A GMP facility keeps a “biography” of every batch, from the raw powder to the final seal. If something goes wrong, they can trace it back to the exact minute it happened.
2. Trust, but Verify: The Power of Third-Party Testing
A company telling you their product is “100% pure” is one thing; an independent lab proving it is another. You should always look for a Certificate of Analysis (CoA) for your specific batch.
What to look for on a Peptidee CoA:
- Purity (HPLC): Aim for 97% or higher. High-Performance Liquid Chromatography (HPLC) acts like a filter, ensuring you get the peptide you paid for and not a vial of filler.
- Identity (Mass Spectrometry): This confirms the “molecular fingerprint” of the peptide. It ensures the BPC-157 you ordered isn’t actually something else entirely.
- The “Nasty” List: The report should show “Pass” or “ND” (None Detected) for:
- Bacterial Endotoxins: Irritants that can cause fevers or inflammation.
- Heavy Metals: Lead, arsenic, and cadmium have no place in your body.
- Residual Solvents: Chemicals like acetonitrile used in the lab must be fully cleared out.
3. Sourcing of Peptide: Don’t Be Fooled by Fancy Labels
Just because a website looks professional doesn’t mean their lab is.
- Pharmaceutical Grade: Even if a peptide is sold for “research,” the synthesis should happen in a pharmaceutical-grade environment.
- Reputation Over Price: If the price seems too good to be true, it likely is. Quality reagents and sterile lab time are expensive. Choosing a supplier with a long-standing track record is an investment in your safety.
4. You’ve Got the Goods—Now Don’t Spoil Them!
Peptides are fragile biological strings. Once they arrive at your door, the clock starts ticking.
- The Deep Freeze: Keep unmixed (lyophilized) powders in the freezer or fridge, away from light.
- The Mix: Once you add bacteriostatic water, the peptide is “activated” and more prone to breaking down. Keep it in the fridge and try to use it within 4 weeks.
- Handle with Care: Never shake a peptide vial! Gently swirl it. Think of it as a delicate snowflake; rough handling can “denature” or break the peptide chain, making it useless.
Peptides Reality Check on Regulations
Most peptides are currently classified as “research chemicals.” This means they don’t have the same FDA “stamp of approval” as your local blood pressure medication. Because the oversight is lower, your standards must be higher. By insisting on GMP standards and third-party CoAs, you are creating your own safety net.
Ready to Optimize Your Peptides?
Navigating the world of peptides can feel overwhelming, but you don’t have to do it alone. Ensuring you have the right protocols and the highest quality sources is what I’m here for.
Would you like me to review the Certificate of Analysis (CoA) for your current supplements or help you create a “Site Rotation Log” for you to help track where you’re injecting each day?
We offer full peptide optimization consults. Let’s chat during our next house call!
You can also submit a new client form to help expedite your visit.
Further Reading and Quality Standards
- FDA: Current Good Manufacturing Practice (CGMP) Regulations – The foundational guidelines for ensuring product quality and safety in the US.
- World Health Organization (WHO): GMP for Pharmaceutical Products – Global standards for manufacturing and quality control.
- PIC/S: Pharmaceutical Inspection Co-operation Scheme – International standards used to harmonize GMP inspections worldwide.
- USP <797>: Pharmaceutical Compounding – Sterile Preparations – The industry standard for the sterile preparation and handling of injectable products.
- ICH Q3C: Impurities – Guideline for Residual Solvents – Scientific guidelines on the safe limits of solvents used during peptide synthesis.
- NCBI: Stability and Storage of Therapeutic Peptides – A scientific look at how temperature and handling affect peptide integrity.
- CDC: Injection Safety Guidelines – Best practices for preventing infection and ensuring one-time syringe use.


